Nanoscale Tungsten Phosphide Prepared by Ammonium Metatungstate: An Efficient Catalyst for Hydrogen Production
- Details
- Category: Tungsten Information
- Published on Sunday, 22 August 2021 14:11
Due to the environmental pollution and climate change cause by burning of fossil fuels, finding a renewable and clean energy alternative is becoming increasingly important. Hydrogen (H2) has high energy density and zero emission, which makes it one of the most promising energy carriers in future. Among various hydrogen production methods, hydrogen evolution reaction (HER) from electrocatalytic water splitting is considered as an ideal eco-friendly strategy to generate hydrogen in a large scale.
Platinum (Pt) and Pt-based noble metals is considered to the best HER catalyst but their high expense and terrestrial scarcity significantly impede their practical applications. Proton exchange membrane-based electrolysis systems. Moreover, they suffer from poor stability resulting from corrosion in low pH media. Hence, It is necessary to find a new stable, non-noble-metal HER catalyst.

Tungsten phosphide (WP) has attracted increasing attention in hydrogen evolution reaction due to its unique catalytic and electronic properties. Tungsten phosphide nanorods (WP2) has been prepared by ammonium metatungstate, which has a great potential as an efficient catalyst for hydrogen production. The result proves it to be excellent catalyst for hydrogen evolution reaction. The synthesis process is as following steps:
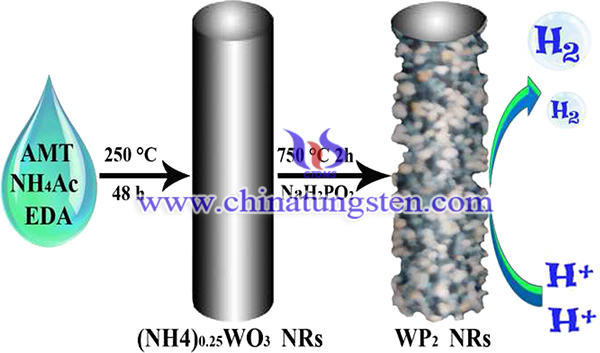
Firstly, 0.167 mmol ammonium metatungstate (AMT) was dissolved in 20 mL of distilled water, followed by adding 18.0 mmol ethylenediamine (EDA) and 10.0 mmol of ammonium acetate (NH4Ac). After completely dissolving under magnetic stirring, the solution was transferred and sealed into a Teflon-lined autoclave with an internal volume of 50 mL, then heating to 250 °C with a ramp of 2.5 °C min−1 and keeping for 48 h in an electric oven. The green product was centrifuged and washed with distilled water and absolute alcohol several times, respectively. Finally, the product was dried in a vacuum oven at 60 °C for 24 h. Then, the previously-prepared (NH4)0.25WO3 NRs and sodium hypophosphite (NaH2PO2) with 1:20 in mole ratio of W/P were placed in two closed porcelain crucibles to heat up to 750 °C with a ramp of 5 °C min−1 and keep at this temperature for 2 h in Ar atmosphere, followed by cooling to ambient temperature naturally under Ar atmosphere.
In conclusion, nanoscale tungsten phosphide have been prepared by ammonium metatungstate, which has a great potential as an efficient catalyst for hydrogen production. The WP2 NRs catalyst exhibits superior catalytic activity and excellent durability in acid (pH 0.3), neutral (pH 7.0) as well as basic medias (pH 14.0), which makes it as an excellent earth-abundant alternative to Pt-based catalyst towards water electrolysis for mass-production of hydrogen fuels. The catalytic activity of catalyst is affected by synthesis temperature and mainly influenced by the oxidation state of tungsten.
- AMT Manufacturer & Supplier, Chinatungsten Online: ammonium-metatungstate.com
- Tungsten News & Prices of China Tungsten Industry Association: www.ctia.com.cn
- Molybdenum News & Price: news.molybdenum.com.cn
- Tel.: 86 592 5129696; Fax: 86 592 5129797; Email: sales@chinatungsten.com



 sales@chinatungsten.com
sales@chinatungsten.com