Synthesis of Tungsten Oxide Nanobricks Using Ammonium Paratungstate
- Details
- Category: Tungsten Information
- Published on Thursday, 29 October 2020 00:41
WO3 is a popular and least bothered an n-type semiconductor with promising electrochemical properties, a high surface area, a small pore-size distribution, with several exciting chemical and physical properties, which have made it a appropriate material for various applications such as catalysts, gas sensors, and energy storage applications[5]. Up till, myriads of chemical and physical preparation methods like thermal evaporation, chemical vapor deposition, the sputtering, and colloidal suspension, have been applied for the synthesis of WO3 nanostructures of different phases and morphologies; nanoparticles, nanofiber, nanorods and nanowires. All reported synthesis methods could be either expensive, time consuming or tedious, with huge hazardous waste as toxic byproducts.
Hydrothermal synthesis has received great consideration because of its simplicity, cost-effectiveness, soft chemical approach, low-temperature processing, and uniqueness in producing controlled shapes/sizes of various nanostructures.
WO3 is a potential sensor material because of its large surface area and unique structural, optical, and electrical properties. However, WO3 as a sensor has endowed its effective sensing operation above 150 °C; not in position of fabricating low-temperature operating sensor devices. Moreover, a high-temperature operation endows limited practical applications, viz., and detection of flammable gases. Several consecutive efforts have developed to reduce the operation temperature of these sensors likewise; the use of metal oxide-based gas sensors to detect various target gases at atmospheric temperature.
Moreover, above drawbacks might overcome by controlling the structure, morphology and surface area of WO3, a facile, simple and low-cost hydrothermal method is used to obtain WO3 NB gas sensors operating at room temperature. The WO3 NB gas sensors approve higher selectivity towards ammonia, than other VOCs such as ethanol, methanol, acetone, and toluene. The sensor for ammonia gas endows an ultra-fast response/recovery time. The schematic of gas sensing mechanism using a band structure change is closely analogous to experimental findings. The WO3 NBs gas sensor has also demonstrated remarkable sensing response for humidity.
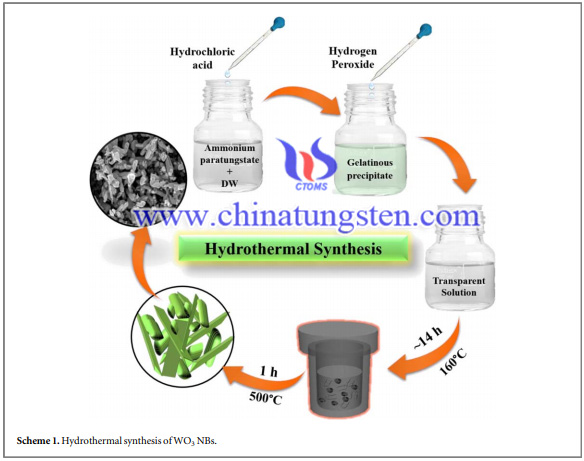
The hydrothermal synthesis of tungsten oxide nanobricks using ammonium paratungstate as the raw material has followed the procedures:
Analytical grade ammonium paratungstate (NH4)10[H2W12O42]·4H2O, 99%), hydrochloric acid (HCl, 37%), and hydrogen peroxide (H2O2, 30% w/w in water)were used as received. In a facile synthesis process, 1 g of ammonium paratungstate was mixed in 95 ml distilled water(Milli-Q water; 18.2 MΩ cm) and concentrated HCl(∼3 ml) and then stirred for a while. The stirring time was depending on the amount of HCl added. After the addition of nearly 2 ml of H2O2, the ammonium paratungstate solution initially turned to a transparent light yellow and then dark yellow by forming gelatinous precipitate, which was evidenced after continuously 1 h stirring to form transparent solution.
A transparent solution was poured in a stainless-steel Teflon-lined autoclave. The hydrothermal synthesis reaction was conducted at a constant temperature of 160 °C for∼14 h (heating rate 5 °C min−1). The whitish powder was taken off, rinsed with distilled water and calcinated at 500 °C for 1 h to increase the crystallinity of the as-obtained WO3 NBs. The solid-state film was fabricated by taking 1 g of polyvinyl alcohol (PVA) in 10 ml of distilled water as a binder, which was kept at 90 °C while constant stirring until the solution became viscous. Then, a few drops of viscous PVA solution was mixed with the WO3 NBs in a pestle and mortar. This mixture was coated onto a soda-lime glass substrate and dried at 300 °C to vaporize the excess water and binder. The obtained film was used as a room-temperature gas and humidity sensor.
In conclusion, mesoporous WO3 NBs were prepared via a simple and cost-effective hydrothermal synthesis method. A film of WO3 NBs was fabricated directly onto a borosil-glass substrate using a doctor-blade method. The gas sensing performance of WO3 NBs sensor was measured for the detection of ammonia and humidity at room-temperature (27℃). The highest response of 75% and ultrafast 8/5 s response/recovery time were obtained at 100 ppm concentration of ammonia. In addition, an excellent humidity response (32%) was displayed by the WO3 NBs sensor at a relative humidity (RH) of 20%. The fast response time (10 s) of humidity sensing is due to the hydrophilic surface of the WO3 NBs can be accounted for a slow H2O molecules adsorption and an ultra-fast recovery time of 8 s; this could be because of the rapid desorption of H2O molecules on the surface of WO3 NBs.
- APT Manufacturer & Supplier, Chinatungsten Online: ammonium-paratungstate.com
- Tungsten News & Prices of China Tungsten Industry Association: www.ctia.com.cn
- Molybdenum News & Price: news.molybdenum.com.cn
- Tel.: 86 592 5129696; Fax: 86 592 5129797; Email: sales@chinatungsten.com



 sales@chinatungsten.com
sales@chinatungsten.com